As the cannabidiol (CBD) market continues to gain steam, global regulators are working to carve out or update legal pathways to market. This makes keeping up—and staying compliant—an ongoing challenge for brands.
Nutrasource Blog
Your (Canadian) CBD Questions Answered
Posted by Jennifer Andrews, M.Sc., MBA, Marketing Director on Thu, May 02, 2019
Tags: Product Marketing, Regulatory, Cannabis
Go to Market in Multiple Countries with One Product Spec
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Tue, Apr 02, 2019
What if you could develop one dietary supplement, with a single product specification, and sell it in multiple countries simultaneously – with no reformulation or regulatory headaches involved?
Tags: Product Marketing, Claims, Regulatory, Concept to Claim, Dietary Supplements/Natural Health Products
Ready to Comply with New Supplement and Nutrition Labels by 2022?
Posted by Amy Mozingo, MS, Director of Operations - GRAS Associates on Tue, Mar 05, 2019
Note: This article pertains to the United States. Products sold elsewhere are subject to different country/region-specific regulations.
Mandatory label compliance deadlines are drawing near. If you sell, or are planning on selling, a dietary supplement or food in the U.S., here is what you need to know about the updates required and how you can comply.
In addition to the changes to nutrition labeling, due for most companies January 1, 2020, FDA has set an official “uniform compliance date” of January 1, 2022 for final food and dietary supplement label regulations issued in 2019 and 2020. This compliance date also applies to the National Bioengineered Food Disclosure Standard.
With so many label updates coming in the near future, will your brand ready?
Tags: Regulatory, Foods & Beverages, Dietary Supplements/Natural Health Products
The Supplement Industry Still Has a Claims Substantiation Problem. Here's How to Fix It
Posted by Douglas Kalman, Ph.D., R.D., CCRC, FACN, Vice President of Scientific Affairs on Wed, Jan 30, 2019
For the past decade, I've written and educated industry about the benefits of substantiating dietary supplement claims, from the rationale of complying with national regulatory rules to remaining ahead of the innovation curve.
But despite policing from the U.S.-FDA and FTC, and the negative media attention surrounding offending brands, the supplement industry still has a substantiation problem. There are far too many instances of companies under fire (or yet to be under fire) for unsubstantiated, misleading, or deceptive claims.
The good news is that this can be fixed with a clearer understanding of what qualifies as substantiation and how companies can benefit from making the investment in quality research. Read on to find out what you need to know.
Tags: Product Marketing, Claims, Regulatory, Concept to Claim, Dietary Supplements/Natural Health Products
Regulatory Trends That Will Shape 2019
Posted by Jennifer Andrews, M.Sc., MBA, Marketing Director on Thu, Dec 06, 2018
The past year was a whirlwind of regulatory change that brought exciting opportunities and new challenges in equal measure.
As we look to another game-changing year ahead, our regulatory and scientific team reviews the top regulatory trends expected to shape health product innovation and marketing in 2019 and beyond.
Here's what to expect...
Tags: Product Marketing, Claims, Regulatory, Cannabis, Foods & Beverages, Dietary Supplements/Natural Health Products
How to Form a CRO Partnership That Delivers Results
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Mon, Oct 15, 2018
The term "partnership" has become a trendy word in the contract research organization (CRO) world. While it's meant to describe a close collaborative relationship between CRO and client, too many organizations claim to be partners when in reality they are only research sites or project-based consultants.
Prop 65 Updates: How to Ensure Dietary Supplement Compliance in California
Posted by Kevin Yan, M.Sc., Director of Analytics & Certifications on Fri, Sep 07, 2018
In the wake of California's updated Prop 65 requirements—which came into force August 30, 2018—many of our clients have approached us with questions regarding compliance.
Tags: Product Testing & Certifications, Regulatory, Dietary Supplements/Natural Health Products
Substantiating Products With No Guidance: The CBD Confusion
Posted by Susan J. Hewlings, Ph.D., R.D., Director of Scientific Affairs on Fri, Aug 17, 2018
While many U.S. states (and, of course, Canada) are approving medical and recreational use of cannabis products, confusion about how these products are regulated continues to grow.
Tags: Claims, Regulatory, Cannabis
Implications of FDA's GRAS and NDIN Policy Changes for Supplements and Medical Foods
Posted by Dr. Richard Kraska, Ph.D., Chief Scientific Officer - GRAS Associates on Tue, Jul 24, 2018
Our regulatory affairs team has regular contact with the U.S. Food and Drug Administration (FDA) for a number of projects. Occasionally we learn about subtle policy shifts regarding Generally Recognized as Safe (GRAS) Notifications and New Dietary Ingredient Notifications (NDIN).
Below is an overview of FDA’s latest updates to GRAS and NDINs which will have important implications for the dietary supplement and medical foods industry in the U.S.
Tags: Regulatory, Dietary Supplements/Natural Health Products
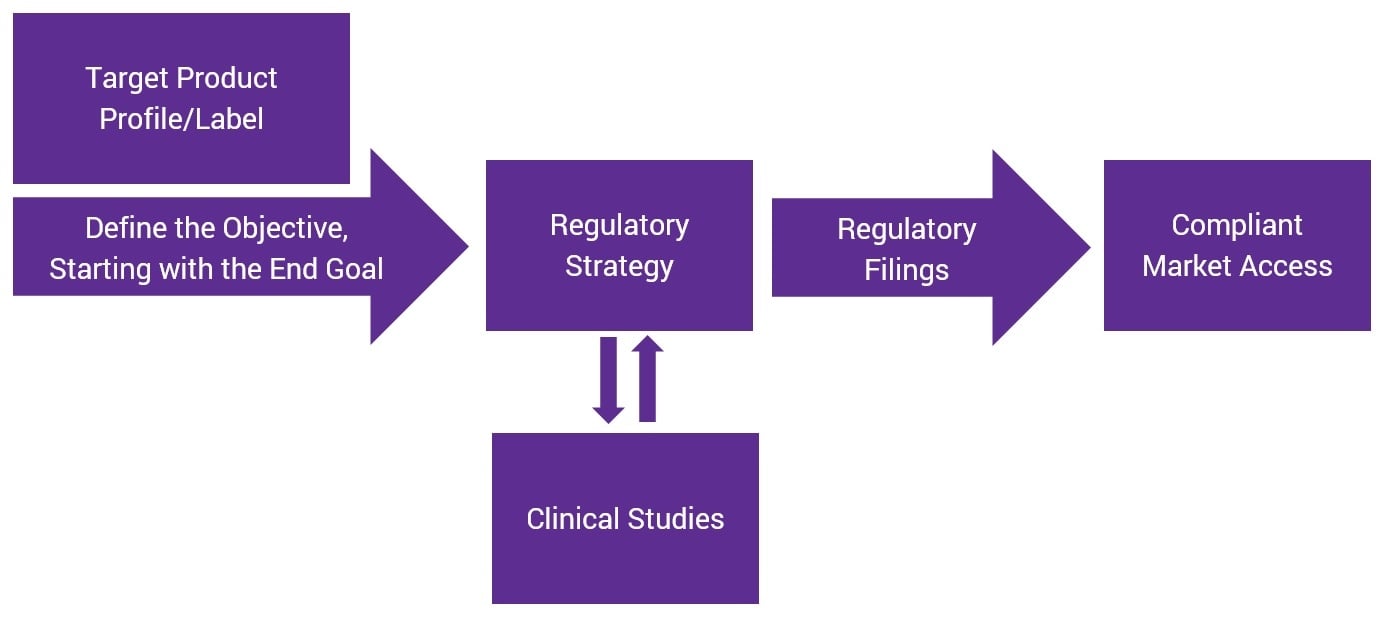
Reverse-Engineering Your Regulatory Strategy for More Efficient Product Development
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Thu, Jul 12, 2018
Regulatory strategy is the backbone of successful product development. Brands that lack a clear regulatory plan at the beginning of the process can face longer times to market and potentially higher costs associated with re-work or failure.
Tags: Clinical Trials, Regulatory, Dietary Supplements/Natural Health Products