The Association of American Feed Control Officials (AAFCO) maintains a list of feed ingredient names and the definitions in the AAFCO Official Publication. This list is central to harmonizing inter-state trade for animal feed and is referenced by international organizations. In late 2024, it was announced that the AAFCO “ingredient definition” process was going to change when the memorandum of understanding between AAFCO and the FDA came to an end. The FDA quickly put forward a draft guidance that has since been finalized for Animal Food Ingredient Consultation (AFIC) process as an alternative pathway, complementing the Food Additive Petition (FAP) and Generally Recognized as Safe (GRAS) notifications that exist currently. Similarly, AAFCO released a draft proposal from Kansas State University (KSU) for a new AAFCO ingredient definition process that does not involve the FDA.
Nutrasource Blog
(Almost) All Roads Lead to AAFCO: A Regulatory Update for Feed Ingredient Manufacturers
Posted by Kaiti Valm, M on Wed, Feb 12, 2025
Tags: Regulatory, ingredients, Animal Health
Dietary Supplements in the U.S.: Labeling Requirements and Structure-Function Claims
Posted by Nutrasource on Tue, Jan 14, 2025
Whether you’re a startup brand introducing your first product or an established brand with many SKUs, the regulatory requirements are the same: companies are responsible for ensuring that their dietary supplements comply with U.S. laws governing safety, quality, and labeling.
The dietary supplement market in the U.S. includes a diverse range of products, including vitamins, minerals, herbal preparations, probiotics, and specialty nutrients. These products fall under the jurisdiction of the Food and Drug Administration (FDA) and are regulated differently than conventional foods or drugs. Unlike some countries, such as Canada or members of the European Union, where pre-market approvals are required, the U.S. system relies primarily on post-market oversight.
Tags: Regulatory, Concept to Claim, Dietary Supplements/Natural Health Products
When it comes to food and supplement labeling, there are significant differences between the US and Canada – even spelling the word “labelling (labeling)” is different. Both markets have their own set of regulations which detail the mandatory content required on a product label. The regulations also give guidance on voluntary content such as claims. It’s important to know which governing bodies have oversight, which regulations apply and how to properly classify your product to make sure you use the right set of regulations from the beginning.
Tags: Regulatory, Concept to Claim, Dietary Supplements/Natural Health Products
Fulfilling GRAS Requirements: Tips for a Successful Manuscript Publication
Posted by Nutrasource on Tue, Oct 22, 2024
Thinking about getting your toxicological results published? Go no further until you read this article.
Tags: Claims, Regulatory, Concept to Claim, market access
Thinking about placing a toxicological test on your novel ingredient? Go no further until you read this article.
Tags: Claims, Regulatory, Concept to Claim, market access
Ensuring Safety: Addressing Contaminants in Food and Dietary Supplements
Posted by Nutrasource on Thu, Jul 25, 2024
You may have a blockbuster food or dietary supplement ingredient, but if the manufacturing process introduces contaminants, it may not be safe to consume. Contaminants may be introduced into a botanical ingredient during growth, such as heavy metals or herbicides from soil or pesticides or antifungal agents applied to plants. Toxins can be produced by plants or be introduced into plants during storage by fungal contamination. Ingredients that are produced by chemical synthesis may be contaminated with solvents or unintended metabolites, and ingredients that are manufactured by microbial fermentation can be contaminated with the microbes or toxins produced by them. Other sources of contaminants include water, processing aids, filters/columns, fermentation media ingredients and packaging materials. Use of Good Manufacturing Practice (GMP), potable water and food grade materials (by U.S. standards) can help reduce levels of contaminants in an ingredient. However, even if these procedures are in place, contaminants may be present in your ingredient.
Tags: Claims, Regulatory, Concept to Claim, market access, Animal Supplements
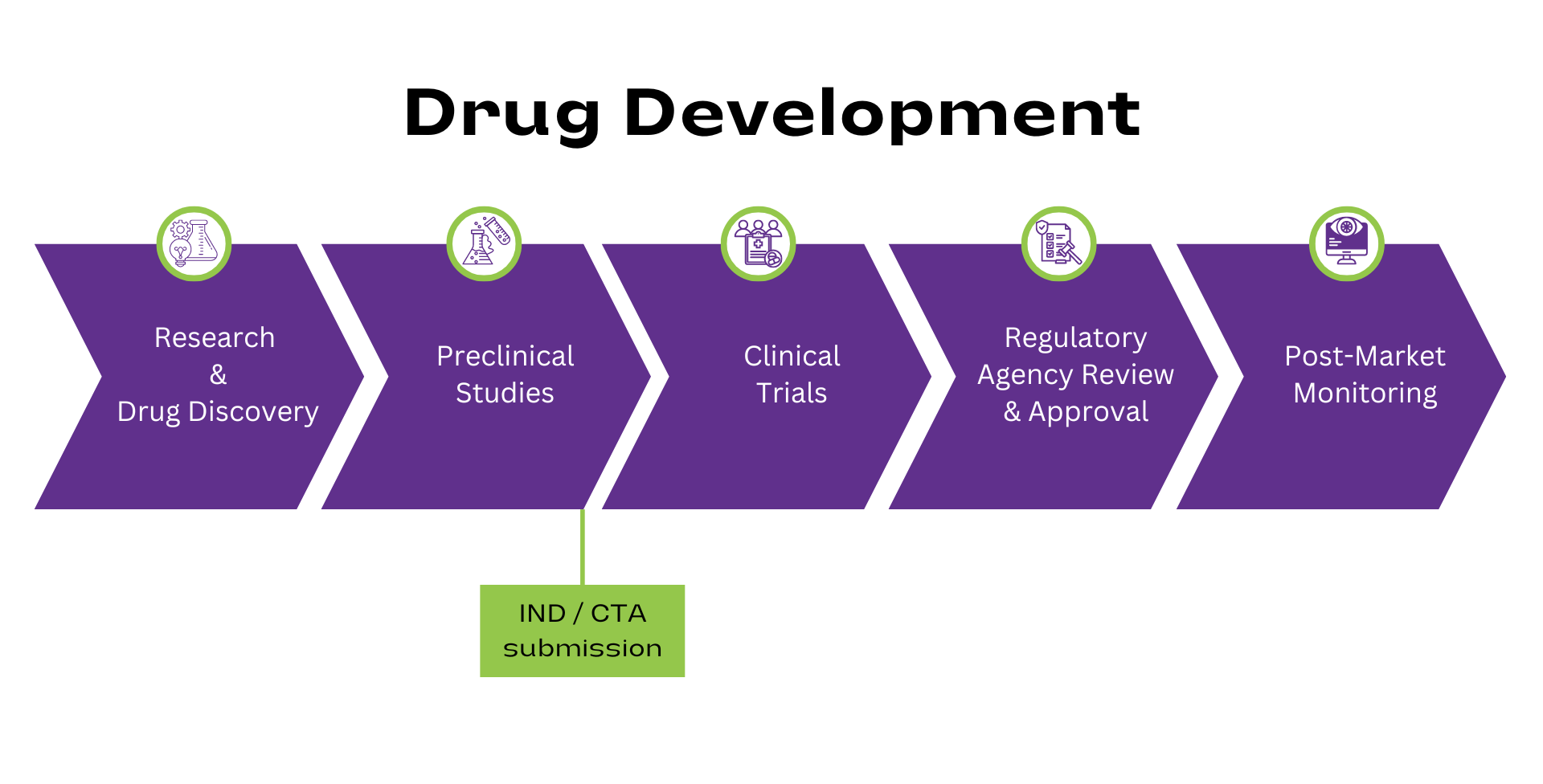
The Importance of Investigational New Drug (IND) Applications and Clinical Trial Applications (CTAs) for Drug Development Strategy
Posted by Nutrasource on Thu, Jun 20, 2024
Drug development is a complex, multi-phase process, involving rigorous testing to ensure safety and efficacy. Prior to drug or biologic testing in human subjects, approval must be obtained from regulatory authorities like the United States (US) Food and Drug Administration (FDA) if the drug or biologic is to be marketed in the US and Health Canada if Canada is the market of interest. In the US, approval is obtained through an Investigational New Drug (IND) application, while in Canada, the application required is a Clinical Trial Application (CTA).
Tags: Regulatory
Marketing Claims for Natural Health Products in Canada
Posted by Tania John, M.Sc. on Fri, May 03, 2024
Natural Health Products (NHPs) are naturally occurring substances that maintain, promote, or restore good health.1 To receive Health Canada approval, an NHP must carry at least one health claim and that health claim must have a health context. For example, “Source of antioxidants for the maintenance of good health.” In addition to the recommended use or purpose statement(s), there are several parameters that form part of an NHP’s Product Licence or Terms of Market Authorization (TMA), including product classification, product representation, composition / ingredients, directions for use, duration of use, risk information, allergen source disclosure, storage conditions, etc.2 Marketing claims can be made around any of these aspects, and while they do not typically appear in the Product Licence Application (PLA) since they lack the required health context, they need to be consistent with the product’s TMA. Product performance, content, comparisons, representations of opinion and authorization, and side effect claims may also be considered marketing language.3 Some examples of acceptable advertising claims are provided in the Table below.
Tags: Product Marketing, Regulatory, Dietary Supplements/Natural Health Products
UNDERSTANDING MASTER FILES
Master Files (MFs) are reference documents that contain proprietary and/or publicly-available data on aspects of drug or Natural Health Product (NHP) safety, efficacy, and/or quality that are submitted to regulatory authorities in support of various application types, including Clinical Trial Applications (CTAs), Investigational New Drugs (INDs), Abbreviated New Drug Applications or Submissions (ANDAs / ANDS), New Drug Applications or Submissions (NDAs / NDS), and Product Licence Applications (PLAs).
MASTER FILES SUBMISSION
MFs are submitted by individuals or companies that develop, manufacture, or supply drugs or NHPs, and are known as the MF Holders. In some jurisdictions the entire MF is strictly confidential between the MF Holder and the regulatory Agency, whereas in others restricted access is granted to Applicants authorized by the MF Holder. Once authorized, the Applicant is allowed to reference the MF in their own submission(s).
The purpose of a MF is to provide sufficient details to the regulatory Agency to satisfy safety, efficacy, and/or quality requirements. The information used in a MF can include specifics about the facilities, processes, and components used in manufacturing, processing, packaging, and storage, without disclosing proprietary information to the Applicant. The MF Holder is responsible for maintaining the MF and ensuring that it is kept up-to-date throughout its lifecycle.
TYPES OF MASTER FILES BY JURISDICTION
In Canada, an NHP-MF serves the same purpose and follows a similar submission process as that of a Drug Master File (DMF). The United States (US) also has DMFs, whereas they are referred to as Active Substance Master Files (ASMFs) in Europe – both regions do not have a MF pathway for dietary or food supplements.
The type of MF needed depends on the regulated category and information intended to be communicated to the regulatory authority. For example, Health Canada and the US Food and Drug Administration (FDA) recognize five (5) different types of DMFs, yet the European Medicines Agency (EMA) only uses one (i.e., ASMF). Health Canada’s Natural and Non-prescription Health Products Directorate (NNHPD) also has a standalone MF specific to NHPs. The table below provides a summary of the different MF types across each of the three jurisdictions.
FIND OUT MORE:
Nutrasource’s NHP & Pharmaceutical Regulatory Sciences team is well-versed in MF dossier preparation and submission to various regulatory authorities.
Our experts can help navigate between MF types and the requirements for each section. Contact us today.
REFERENCES:
4. Drug MF https://www.fda.gov/drugs/guidances-drugs/drug-master-files-guidelines
5. Drug MF https://www.fda.gov/drugs/forms-submission-requirements/drug-master-files-dmfs
6. US Dietary Supplement https://www.fda.gov/news-events/press-announcements/fda-updates-draft-guidance-premarket-safety-notifications-dietary-supplement-industry
7. Drug MF https://www.fda.gov/media/85079/download
8. Active Substance Master File https://www.ema.europa.eu/en/documents/report/final-guideline-active-substance-master-file-procedure-revision-4_en.pdf
Tags: Regulatory, Dietary Supplements/Natural Health Products
VHPs in Canada: A Growing Market with a Regulated Pathway
Posted by Nutrasource on Fri, Aug 11, 2023
Tags: Claims, Regulatory, Concept to Claim, market access, Animal Supplements








.jpg)
.png)
