Study designs need to support the final claims including marketing claims. Risk management is a key aspect of formulating a robust clinical development program and may include proof of concept studies such as dose ranging or multiple dose studies and pilot studies, prior to engaging in pivotal clinical studies to determine evidence of effect and variability around changes resulting from supplementation with the investigational product.
Nutrasource Blog
Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery
Recent Posts
Understanding the Differences Between Phase I, II, III, and IV Clinical Trials
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Thu, Dec 03, 2015
Tags: Clinical Trials, Dietary Supplements/Natural Health Products
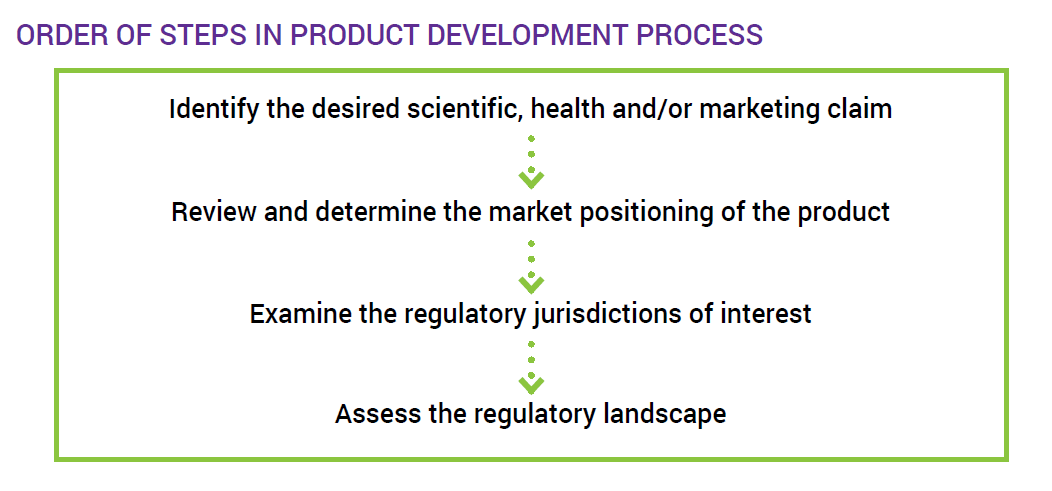
Start with the Product Claim When Developing a Dietary Supplement
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Thu, Nov 12, 2015
There are many questions that must be asked, and consecutive steps planned, in order to successfully develop and launch a dietary supplement. These questions include:
Tags: Claims, Concept to Claim, Dietary Supplements/Natural Health Products
Is Your Dietary Supplement Commercialization Plan on Target?
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Wed, Jul 22, 2015
Clinical research has become a fundamental step in the development of a dietary supplement. Nutraceutical companies are investing more in clinical research than ever before to support health and marketing claims and gain a competitive advantage in a saturated marketplace.
Tags: Clinical Trials, Pharmaceuticals, Claims, Concept to Claim, Dietary Supplements/Natural Health Products
Benefits of Conducting Clinical Research in Canada
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Tue, Jun 02, 2015
Clinical research in Canada has been declining year over year since 2010. Fluctuations are sometimes seen due to the changing strength of the Canadian versus American dollar as the costs are typically quite similar between countries. Another factor in the recent downturn has been partially credited to Sponsors and contract research organizations (CROs) placing studies in foreign countries or being misinformed that data must be produced in the country they are marketing in. These factors have led to CROs and sites to move to, or open operations in, other countries to adapt.
Tags: Clinical Trials, Dietary Supplements/Natural Health Products
Can Your Dietary Supplement Claims Stand Up to the FTC?
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Fri, Feb 06, 2015
The verdict is in on one of the two high-profile dietary supplement industry cases before the courts this year. The decision from the U.S. Court of Appeals for the D.C. Circuit upheld the Federal Trade Commission's (FTC) charges that POM Wonderful and POMx ads were deceptive. The ads claimed that the product could treat, prevent or reduce the risk of heart disease and prostate cancer, but lacked clinical evidence to substantiate such claims.
The decision led to questions around how much clinical evidence is required to support a health claim. How is a company promoting the benefits of natural health products supposed to navigate the regulatory landscape when even the guidances are not clear?
Tags: Product Marketing, Clinical Trials, Claims, Dietary Supplements/Natural Health Products