Content contributed by
.png?width=560&height=78&name=Article%20Contribution%20-%20hubspot%20size%20(9).png)
.png?width=560&height=78&name=Article%20Contribution%20-%20hubspot%20size%20(10).png)
Decentralized clinical trials (DCTs), also termed “virtual trials”, “site-less trials”, or “direct-to-participant trials”, are defined as the execution of clinical trials with the help of virtual visits or mobile healthcare providers, using technologies and methods that differ from a traditional clinical trial setup. DCTs can refer to both fully DCTs (virtual) and hybrid DCTs. In a fully decentralized clinical trial, participant recruitment, consenting, administration of study medication, collection of data, etc. will be done with no in-person study visits. In hybrid DCTs, most of the study procedures and visits are performed remotely, but the study participants may need to be on-site for some procedures such as physical examinations, sample collection, complex treatments, etc. The complexity of a clinical trial and the target population are key parameters that dictate whether to opt for a fully DCT, a hybrid DCT, or classic on-site trial approach to achieve the best results.
Virtual technologies
DCTs have also revolutionized the development and implementation of digital health technologies (DHTs) to support clinical research and collect real-time data remotely from the study participants. DCTs require various virtual tools and setups such as virtual visits, wearable medical devices, phone apps, etc., which are all examples of DHTs.

Are decentralized trials really the future of clinical trials?
There are some advantages of DCTs over traditional clinical trials. Conducting virtual visits can reduce the participant and caregiver burden (e.g., travel, parking, waiting time for an appointment). Wearable devices may also be utilized to monitor vital signs such as heart rate, respiration, blood pressure, and temperature remotely, which could help in early detection of treatment-related adverse effects. Such devices can also track activities of daily living, such as sleep duration and quality, step count, time in sedentary positions vs. walking/exercising, etc. DCTs can cover a broader geographical area and can target wider demographic groups. Using DHTs can help study participants and caregivers with study alerts and reminders, scheduling of study visits, and remote data collection. The US FDA has recently proposed that DCTs might reduce barriers and cater to a wider range of participants by reducing the costs and commitments required for study participants to take part in a clinical trial.
Potential limitations of DCTs
In on-site clinical trials, the study products are shipped to a clinical trial site and distributed to study participants by trained site staff. In DCTs, the study products are shipped directly to the study participants. It may be a challenge to monitor and maintain the proper temperature for study products during transit, which could jeopardize stability and thereby have a negative consequence on study data. Additionally, the protection of privacy and health information is another challenge in DCTs with the use of DHTs. The solicitation, collection, and secure storage of study participant health information requires a thorough understanding of various complex regulatory requirements that vary by region and systems. Identity verification, secure data access and training are critical to addressing this complexity. Study participants need thorough training on the use of technologies for data entry and data capture, including wearable devices and phone applications. The uninterrupted data collection from the DHT gadgets will require a proper cellular plan or wi-fi connection and availability of an IT support team to troubleshoot any technical issues and answer participants’ questions. At the center of technology is also the validity of the software and devices and compliance with federal regulations and international guidelines on clinical research data.
Misconceptions
There are some misconceptions in the dietary supplement industry that DCTs are less costly than traditional on-site trials. While there are benefits to the participant burden and potential decrease in recruitment timelines, there is a shift in labour required for the oversight and quality of the project which is focused more heavily on proper design, deployment and support of software and devices, and the costs for the software platform licensing and devices themselves, which offsets any time savings and costs a site may incur. Further, the costs associated with site staff remain nearly the same as the terms “virtual” and “site-less” are misleading. In these trials, there is still a requirement for site staff to support a DCT by interacting with the participants, performing follow-ups, managing medical oversight, being available for questions, etc. While there may be cost savings for studies such as observational trials, trials requiring thousands of participants, as well as for trials focused on rare indications/diseases, these cost savings are negligible in the dietary supplement industry where recruitment is less challenging due to the nature of the simpler study designs, smaller sample sizes, and more inclusive target populations.
Conclusion
The regulatory bodies need to work together to develop harmonized policies to support DCTs, ensure data integrity and protect sensitive health information of participants involved in the research. Virtual medicine and technologies have gained popularity over the past few years and are now being practiced and used at a larger scale. DCTs are becoming another tool in the clinical research industry alongside traditional clinical trials requiring brick-and-mortar sites, each with their own place and purpose. The successful execution of a decentralized clinical trial is tremendously dependent on the design, operational planning, and continuous oversight of the study. Collaborating with an experienced Contract Research Organization from trial conception is the key to ensuring that the study meets the highest standards of research.
CONTACT US
Exploring options for your clinical trial? Reach out to our team who would be glad to connect and help answer any questions you may have.
RELATED CONTENT

-2.png?width=657&height=183&name=Related%20Content%20-%20for%20website%20pages%20(1)-2.png)

.jpg?width=1792&height=1344&name=Drug%20Submission%20Flowchart%20(6).jpg)

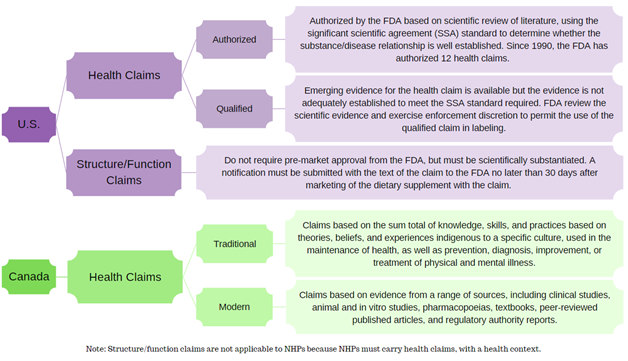
.png?width=1080&name=Claims%20dos%20and%20donts%20(1).png)
-1.png?width=711&height=197&name=Related%20Content%20-%20for%20website%20pages%20(1)-1.png)

.png)


