Whether you are launching a new product or looking to substantiate a novel claim, strong science is crucial to the success of your health product. In Canada, many ingredients have been “pre-cleared” for specific claims at particular doses. Monographs give guidance and provide substantiation for these ingredients, so you don’t have to.
Nutrasource Blog
Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery
Recent Posts
How to Validate Your Products Through Clinical Research
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Thu, Feb 13, 2020
Tags: Clinical Trials, Dietary Supplements/Natural Health Products
Top 3 Reasons to Conduct Clinical Research in Canada
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Tue, Aug 20, 2019
Clinical research is an important investment for the entire supply chain, from ingredient companies to brands selling finished health products. Primary research serves many purposes, and is most commonly used to validate efficacy, safety, and substantiate claims that increase the impact of a company’s marketing messages.
Tags: Clinical Trials
Go to Market in Multiple Countries with One Product Spec
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Tue, Apr 02, 2019
What if you could develop one dietary supplement, with a single product specification, and sell it in multiple countries simultaneously – with no reformulation or regulatory headaches involved?
Tags: Product Marketing, Claims, Regulatory, Concept to Claim, Dietary Supplements/Natural Health Products
How to Form a CRO Partnership That Delivers Results
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Mon, Oct 15, 2018
The term "partnership" has become a trendy word in the contract research organization (CRO) world. While it's meant to describe a close collaborative relationship between CRO and client, too many organizations claim to be partners when in reality they are only research sites or project-based consultants.
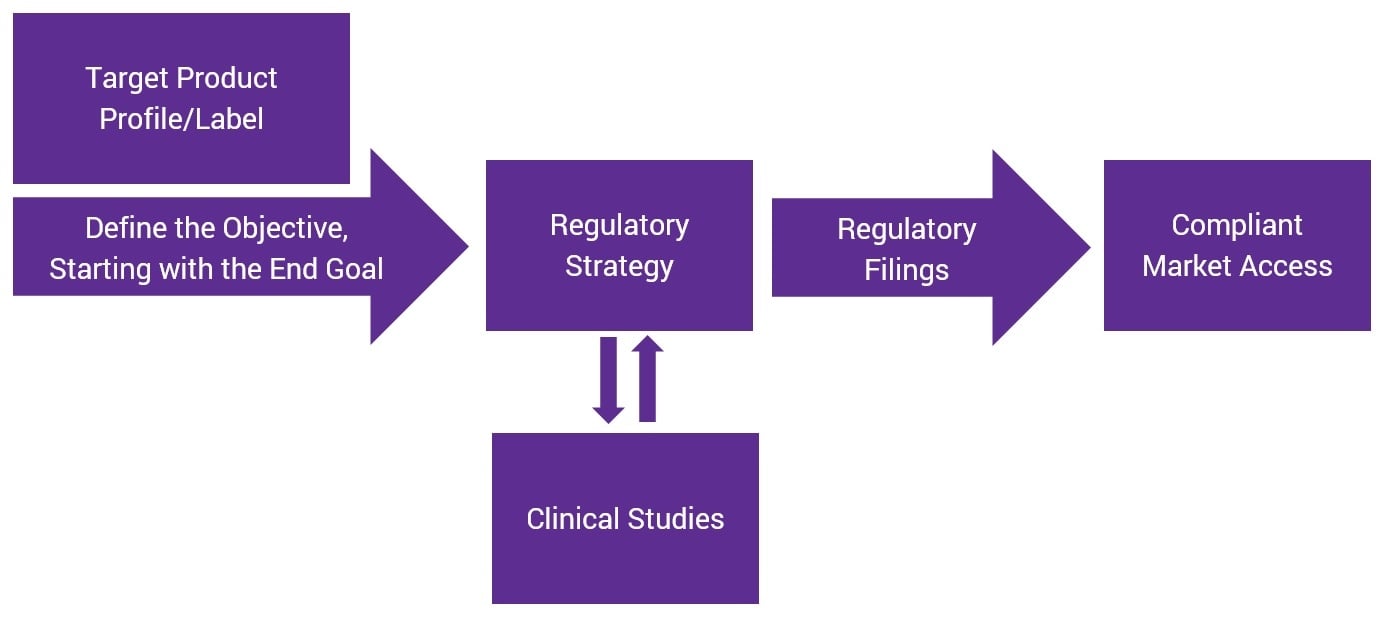
Reverse-Engineering Your Regulatory Strategy for More Efficient Product Development
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Thu, Jul 12, 2018
Regulatory strategy is the backbone of successful product development. Brands that lack a clear regulatory plan at the beginning of the process can face longer times to market and potentially higher costs associated with re-work or failure.
Tags: Clinical Trials, Regulatory, Dietary Supplements/Natural Health Products
What to Expect When Conducting Dietary Supplement Clinical Trials
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Tue, Mar 20, 2018
One of the most common questions we hear from clients early in the product lifecycle is: "How do I let my customers know our product is safe, and that it works?"
Tags: Clinical Trials
Understanding the Clinical Trials Pathway for Probiotics
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Thu, Jan 04, 2018
Making the decision to conduct a clinical trial can be daunting. Many complex steps are involved in getting a study off the ground, including designing the study, assessing risk, and determining whether a trial will achieve return on investment.
Tags: Probiotics
Planning for Risk Mitigation Is Key to a Successful Clinical Research Strategy
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Fri, May 05, 2017
Investing in clinical research is necessary to establish a product’s safety and efficacy. Whether this is your first clinical study on your product or you’ve conducted a number of clinical trials to substantiate claims for your dietary supplement, begin with the understanding that there are risks of all shapes and sizes involved and that the project manager’s first order of business is to eliminate or mitigate as many as possible.
Tags: Clinical Trials
Managing Risk in Probiotic Stability
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Tue, Apr 11, 2017
A comprehensive clinical testing program is essential to the success of virtually all health products. Writing in the International Probiotics Association blog, Josh Baisley, associate director of clinical trials for Nutrasource, notes that ensuring the stability of a probiotic for the duration of clinical studies is a vital aspect of analyzing the outcome.
Tags: Clinical Trials, Probiotics
Demystifying Clinical Study Designs for Probiotics
Posted by Josh Baisley, B.Sc., Vice President, Clinical Design & Delivery on Wed, Oct 12, 2016
As the probiotic segment continues to trend upward, more companies are investing in well-designed clinical trials to support efficacy.
One area that has presented substantial learning opportunities, and has helped shape the future of probiotic clinical trials, is the study of antibiotic associated diarrhea (AAD) and probiotic intake.
Tags: Clinical Trials, Probiotics, Dietary Supplements/Natural Health Products

-1.png)